 The resulting compounds are called ionic compounds. Because opposite charges attract (while like charges repel), these oppositely charged ions attract each other, forming ionic bonds. Those that lose electrons become positively charged, and those that gain electrons become negatively charged. Because some atoms will lose electrons and some atoms will gain electrons, there is no overall change in the number of electrons, but with the transfer of electrons the individual atoms acquire a nonzero electric charge. One way is the transfer of electrons between two atoms until both atoms have octets. There are two ways for an atom that does not have an octet of valence electrons to obtain an octet in its outer shell. This useful rule of thumb is called the octet rule, and it is a key to understanding why compounds form. Chemists have concluded that atoms are especially stable if they have eight electrons in their outermost shell. What else do the noble gas elements have in common? Except for helium, they all have eight valence electrons. /atom-artwork-172594580-58b600b53df78cdcd83c309e.jpg)
These elements-helium, neon, argon, krypton, xenon, and radon-do not form compounds very easily, which suggests that they are especially stable as lone atoms. What causes atoms to make a chemical bond with other atoms, rather than remaining as individual atoms? A clue comes by considering the noble gas elements, the rightmost column of the periodic table. Chemical bonds are formed when electrons in different atoms interact with each other to make an arrangement that is more stable than when the atoms are apart. 3.1 Two Types of BondingĪtoms can join together by forming a chemical bond, which is a very strong attraction between two atoms. We will see additional examples of such differences in section 3.3 of this chapter covering “Covalent Bonding and Simple Molecular Compounds”, as we consider how atoms combine to form compounds. Nevertheless, the compound has properties completely different from either elemental sodium (a chemically reactive metal) or elemental chlorine (a poisonous, green gas). Table salt, as we have seen, consists of only two elements: sodium and chlorine. Compounds can be very complex combinations of atoms, but many important compounds are fairly simple. There are only 118 known chemical elements but tens of millions of known chemical compounds. 3 Covalent Bonding and Simple Molecular Compounds Covalent Bonds Electron Sharing Covalent Bonds Between Different Atoms Multiple Covalent Bonds 3.4 Chapter Summary Ionic Compounds Covalent Compounds 3. 2 Ions Electron Transfer Lewis Diagrams 3. This text is published under creative commons licensing, for referencing and adaptation, please click here. For the interactive PDF, adobe reader is required for full functionality. 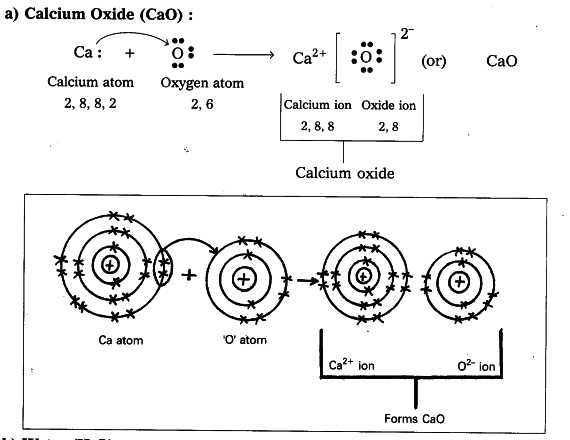
It's thought that another one of these magic numbers lies just beyond the range of currently known elements, which means we could potentially discover some new elements that are relatively stable and extremely massive.This content can also be downloaded as a PDF file. There are believed to be certain numbers/ratios that are inherently more stable. 
Hence, larger atoms become more unstable. The ratio of neutrons to protons gradually increases to try to compensate, but this results in the neutrons getting more unstable themselves, resulting in more opportunity for weak forces to destabilize them. With all of that to consider, as you get larger and larger nuclei with more protons, the electromagnetic forces increase, as well as the distances in the nuclei, putting more strain on the nuclear forces holding it all together. Neutrons will help add to this nuclear force, but they tend to become unstable when they get into too great of concentration and the weak force will cause them to decay into protons. Fusion allows these protons to get close enough together for the stronger, yet short ranged, nuclear force to overpower the electromagnetic force. The electromagnetic force of the protons wants to push them away from each other. There are several forces at work inside the nuclei of atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
